Bonding occurs because it is motivated by the noble gas configuration/octet rule—which is a more stable and a less energy state—because of an increase in the entropy of the universe! The unstable valence electrons want to either leave the atom they are in, or try to attract some electrons from others in order to get a full orbital. Bonding also occurs because it helps to reach the most favorable energy flow, the most favorable structure—symmetry and dipole moments, and the concentration of the reactants. Thermodynamics,the study of the flow of energy in the universe, also plays a big role. First and foremost, you should know that the universe if full of energy! There is practically energy everywhere. There are two main laws of thermodynamics. The first being that energy is always conserved. It cannot be created or destroyed. The second law states that through the amount of energy transfered, a little amount of energy gets lost to the universe which thus contributes to the increase in entropy—amount of disorder in the universe. Bond Energy is the energy required to break a bond as well as the energy required to release bonds—break bonds. Energy is absorbed during endothermic reactions and released during exothermic reactions.
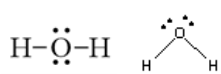
Polarity is one of the biggest bonding theories. It basically determines whether the bond is non-polar, polar, or ionic. First, what exactly do all those mean? Well, a non-polar bond is when the bonding electrons are shared equally—like diatomic halogen molecules. A polar bond would be the opposite, so its when the bonding electrons are shared unequally. Ionic bonds are the electrostatic forces that hold ions together in ionic compounds. To figure this out, you calculate the delta EN (difference in the electronegativities) and then just identify it. If the delta EN = 0 - 0.4, then the bond is non-polar covalent. If the delta EN = 0.4 - 1.9, then the bond is polar covalent. If the delta EN = 1.9 or above, then the bond is definitely ionic. Another theory is the Valence Shell Electron Pair Repulsion (VSEPR). It basically says to remember the repulsions each bonding pair and each lone pair gives to each other. When this theory is used when drawing lewis structures, the structure is more accurate and a much higher possibility. For example, the first image above, with the straight dashed lines—which indicate bonding pairs, would be wrong according to the VSEPR. It would be wrong because the two lone pairs on the oxygen need to be placed differently which will then have an affect on where the hydrogen atoms go. So the perfect example of the VSEPR would be the second image above, with the slanted dashed lines—which indicate bonding pairs. Hybridization is also another bonding theory. To understand this, just remember that structure changes function. An element alone and the same element combined with others will NOT have the same functions and properties. For example, Na (Sodium) alone makes huge explosions and can cause great damage. However NaCl (table salt) does not. When sodium is combined with chlorine, its function completely changes as it no longer causes damage but gives taste to the food we eat.
*****Please note that these models are not perfect as they are just based on theories. Through the theories scientists have developed thus far, these
structures can be assumed to be true.*****
structures can be assumed to be true.*****
Challenger Yourself: Poll
Understand everything going on through reading the above page and then in your own words, try to answer 'Why Does Bonding Occur?' Make sure to use keywords in your answer, like the bolded ones above, to cover all of the important information.
The goal is to use your answers to help make this website much better. Your answers will tell how useful the information is and whether or not it actually helps you. So in a way, you are just helping the creater(s) of this website do a better job in trying to make the information more understandable.
The goal is to use your answers to help make this website much better. Your answers will tell how useful the information is and whether or not it actually helps you. So in a way, you are just helping the creater(s) of this website do a better job in trying to make the information more understandable.
